Category: Fda
My son is fighting for his life. The FDA doesn’t seem to care.

I’ve been fighting Duchenne muscular dystrophy for 40 years. My brothers Angelo and Antonio died from it at ages 20 and 22, respectively. Antonio died in 2015, when my son, Ryu, was barely a toddler and had already been diagnosed with the same terminal illness.
My childhood memories are of praying for my brothers, caring for them with my mother, and Mom taking all five of her kids to church almost every day. I always asked God to heal my brothers, and — after Ryu was born — I added him to those prayers.
I’ve been saying the same prayer for help and to be able to lend my voice for over 40 years.
But I also went to God with another prayer — I asked that He would open the door that allowed me to share our family’s story. I didn’t know what that looked like, or when it would come, but I trusted in it.
This year, that prayer was answered when I was asked to speak out not just on behalf of my brothers and son, but for every family that feels isolated because of a terminal rare disease.
I visited Washington, D.C., to share my story with lawmakers from both parties as well as patient advocates and to ask them to push the Food and Drug Administration to stop standing in the way of drugs like Elevidys, the only gene therapy treatment for my son’s illness.
The advocacy worked. I can’t say how much my own small voice, speaking up for the first time, helped, but so many people speaking out made a difference.
The first indicator was when the Center for Biologics Evaluation and Research Director Dr. Vinay Prasad announced his resignation from the FDA just a week later — he leaves this month. Prasad blocked treatments, with the support of FDA Commissioner Marty Makary, that could have helped kids like Ryu all across the country to live.
RELATED: Trump is keeping his word on health care costs
 Anna Moneymaker/Getty Images
Anna Moneymaker/Getty Images
I’m just a mom. But we recently celebrated Easter, where a carpenter saved the world. He overcame the establishment of His time, which was willing to throw the vulnerable and sick to the side. He fell, but He didn’t falter — I hope to follow His example.
As we were approaching Holy Thursday this year, Ryu was having a hard evening. He needed his Bipap machine to help his lungs function, as he so often does. But he looked at me — my 14-year-old wheelchair-bound boy who is the happiest kid I know — and said, “Mom, this sucks. But what you’re doing makes it a lot easier.”
My story may not matter to FDA Commissioner Makary, who seems to have forgotten about Ryu and thousands of other kids like him. But God sees every hair on our heads. He named us before our parents knew us. And sometimes, like Gabriel told the prophet Daniel, prayers are answered long before we see their fruition.
I’ve been saying the same prayer for help and to be able to lend my voice for over 40 years. To the world, Antonio and Angelo may be long deceased, but they are the foundation for how my husband and I have cared for Ryu. And God has allowed me to carry their stories from my home in El Paso to our nation’s capital.
Commissioner Makary and Dr. Prasad may have forgotten that their job is to save lives, but God seems to have different plans. He’s just getting started with me in spreading His good news, and so far it has been amazing.
But I’m also not surprised, because I knew God would take care of it all.
Editor’s note: This article was originally published in the Christian Post.
FDA Wants Pause On Landmark Abortion Pill Suit. Pro-Lifers Say It Will Cost Lives

 The FDA hinted it does not plan to weigh on the safeguards stripped under Biden until its mifepristone review is complete.
The FDA hinted it does not plan to weigh on the safeguards stripped under Biden until its mifepristone review is complete.
The FDA is undermining a culture of life inside and outside the womb

Last Friday during the annual March for Life, President Trump delivered a pledge to the nation: His administration stands for the “infinite worth and God-given dignity of every human life.” Vice President JD Vance’s remarks at the rally were just as clear: We must “build up that culture of life” and “cannot be neutral. Our country cannot be indifferent about whether its next generations live or die.”
Vance and Trump were primarily talking about the unborn. But their principles clearly include providing the right to life — as well as health and safety — for all citizens, especially the most vulnerable among us.
We have entire policies at the FDA dedicated to making it more difficult for children inside and outside the womb to live the lives they deserve.
Unfortunately, these principles have been undermined by a few key officials at the Food and Drug Administration, and not just for unborn children. Thousands of kids with rare diseases have seen valuable treatments slowed or even halted since last summer, thanks to FDA Commissioner Marty Makary and Chief Medical Officer Vinay Prasad.
As one of the oldest living Americans with spina bifida (I celebrate my 60th birthday this year), I understand the value of providing children with rare and fatal diseases the ability to improve or even extend their lives from a personal, policy, and political perspective. I took that knowledge into the first Trump administration as the commissioner of the Administration on Disability at the Department of Health & Human Services. Today, I’m deeply concerned by what Makary, Prasad, and — at times — Health & Human Services Secretary Robert F. Kennedy Jr. have done to reduce children’s ability to live the full length of their God-given lives.
Those concerns were why I raised the alarm when RFK Jr. was going through his Senate hearings a year ago. He had been openly supportive of abortion on the presidential campaign trail, but I and other concerned pro-life advocates were told that he would have plenty of pro-lifers around him and that people would become policy. They were right: People did become policy, but not the way we had hoped. Now, we have entire policies at the FDA dedicated to making it more difficult for children inside and outside the womb to live the lives they deserve.
Last October, the FDA outraged pro-life warriors across the country by approving a cheaper version of mifepristone, one of the most prevalent and notorious abortion drugs on the market. Women can have these drugs dropped off in their mailboxes and have abortions in the “comfort” of their own homes. The pro-abortion Guttmacher Institute estimates there were over 640,000 chemical abortions in 2023 — 63% of the total abortions that year.
In 2026, there will be even more.
That number, troubling enough on its own, understates the problem because it doesn’t account for the injuries these drugs inflict on the women who take them. One devastating fact I have learned in my advocacy for people with disabilities is the particular hazard the abortion pill presents for women who use wheelchairs or otherwise live with limited mobility. Any drug that causes blood clots — and abortion drugs definitely do — will be a deadly danger to people who have limited mobility.
FDA Chief Medical Officer Vinay Prasad is similarly problematic for those who support protecting life. He not only supports legalized abortion, but since his appointment in mid-2025, Prasad has held up the production of drugs and treatments that would make real differences in the lives of kids who suffer from rare diseases like Sanfilippo syndrome and Duchenne muscular dystrophy.
In 2018, Prasad opposed the Trump “right to try” doctrine, through which hundreds of patients have seen amazing results from drugs still in their experimental stages or through off-label usage. That number could be higher if Prasad’s red tape weren’t keeping effective drugs in “pre-approval” limbo.
RELATED: No, President Trump: The sanctity of life is not ‘flexible’
 Photo by Kyle Mazza/Anadolu via Getty Images
Photo by Kyle Mazza/Anadolu via Getty Images
At HHS, the buck stops with RFK Jr. But ultimately in our government, the buck stops at the Oval Office. Trump and Vance recommitted to supporting life on Friday, and that commitment must be consistent throughout the administration. The FDA’s actions against the unborn and children with disabilities and rare diseases threaten to undermine what should be a slam dunk for Trump’s pro-life legacy.
In short, HHS and FDA appointees should be defending life, not quietly undermining it. Vance and Trump can make that happen.
FDA finally admits COVID-19 vaccine killed kids: ‘This is a profound revelation’
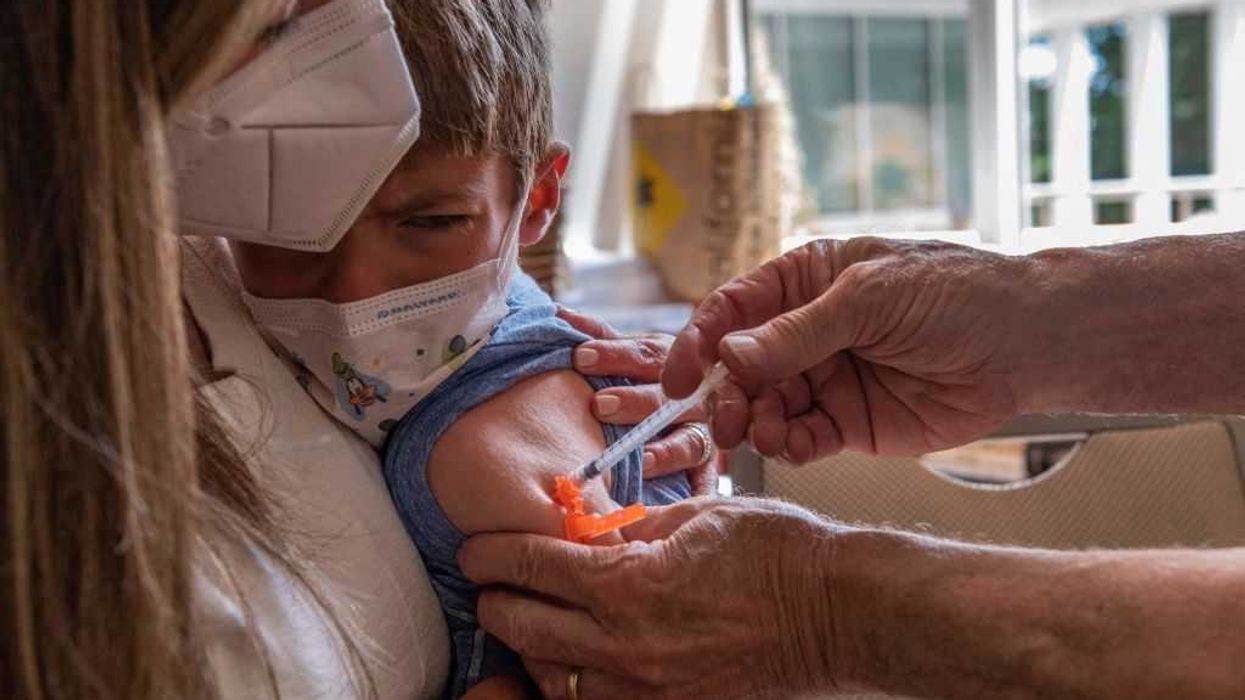
Millions of Americans across the country were told during the pandemic to offer up their arms for the COVID-19 vaccines — the first-ever mRNA vaccines approved by the U.S. Food and Drug Administration — if they wanted to keep their jobs, eat in public, stay in school, or visit their loved ones.
Government officials, the establishment media, and pharmaceutical representatives claimed that the vaccines were “safe and effective.” Those who dared to suggest otherwise about the experimental drugs that were making liability-shielded vaccine manufacturers record profits were often attacked and censored.
Months after the Department of Health and Human Services concluded that “mRNA technology poses more risks than benefits for these respiratory viruses,” the Food and Drug Administration admitted in an internal letter that the COVID-19 vaccines killed numerous children.
‘Healthy young children who faced tremendously low risk of death were coerced.’
Dr. Vinay Prasad, chief medical officer at the FDA and director at the Center for Biologics Evaluation and Research, noted in an email to staff on Friday that FDA Office of Biostatistics and Pharmacovigilance career staff “have found that at least 10 children have died after and because of receiving COVID-19 vaccination.”
In the email, which was reviewed by multiple publications and shared online by the Washington Post, Prasad indicated that the OBPV performed an analysis of 96 deaths between 2021 and 2024 and concluded “that no fewer than 10 are related. If anything, this represents conservative coding, where vaccines are exculpated rather than indicated in cases of ambiguity. The real number is higher.”
“These deaths are related to vaccination (likely/probable/possible attribution made by staff). That number is certainly an underestimate due to underreporting, and inherent bias in attribution,” wrote Prasad. “This safety signal has far-reaching implications for Americans, the U.S. pandemic response, and the agency itself.”
RELATED: Pfizer COVID shot sales plummet after Trump administration ends universal recommendations
 Photo by SAUL LOEB/AFP via Getty Images
Photo by SAUL LOEB/AFP via Getty Images
Despite the strong improbability of a healthy child getting seriously ill from COVID, former National Institute of Allergy and Infectious Diseases Director Anthony Fauci, former Centers for Disease Control and Prevention Director Rochelle Walensky, and other health officials championed injecting kids with the novel vaccines.
On Nov. 2, 2021, then-President Joe Biden’s health officials gave final approval to Pfizer’s COVID-19 shot for kids ages 5 to 11. Biden said at the time, “It is a major step forward for our nation in our fight to defeat the virus.”
COVID-19 vaccination for children younger than 5 began across the U.S. in June 2022.
“These vaccines are safe, highly effective, and will give parents the peace of mind of knowing their child is protected from the worst outcomes of COVID-19,” said Biden.
Prasad noted in his Friday letter that despite evidence that the COVID-19 vaccine put boys and young men at great risk of myocarditis, Biden health officials “did not quickly attempt mitigation strategies such as spacing doses apart, lowering doses, omitting doses among those with prior COVID-19.”
Myocarditis is inflammation of the heart muscle that can manifest as various symptoms, including heart palpitations, chest pain, fainting, and weakness, and can also cause fatal cardiac arrest.
“Worse, the FDA delayed acknowledgement of the safety signal until after it could extend marketing authorization to younger boys 12-15,” continued Prasad. “Had the acknowledgement come early, these younger boys, who likely did not require COVID-19 vaccination, may have chosen to avoid the products.”
The FDA’s chief medical officer stressed that the OBPV’s finding that the COVID vaccine contributed to the deaths of children amounted to “a profound revelation.”
“For the first time, the U.S. FDA will acknowledge that COVID-19 vaccines have killed American children,” continued Prasad, whose agency revoked emergency-use authorization for COVID vaccines earlier this year. “Healthy young children who faced tremendously low risk of death were coerced, at the behest of the Biden administration, via school and work mandates, to receive a vaccine that could result in death. In many cases, such mandates were harmful.”
Peter Marks, Prasad’s predecessor, complained to the New York Times about the “political tone” of Prasad’s letter and noted, “I would not be surprised if the attributions turn out to be debatable, as these cases are often quite complex.”
FDA commissioner Dr. Marty Makary said in a “Fox & Friends” interview on Saturday that his agency would no longer “rubber-stamp things with no data,” adding that such a “mockery of science” was alternatively “the M.O. in the Biden administration with the eternal COVID booster approvals for young, healthy kids.”
Like Blaze News? Bypass the censors, sign up for our newsletters, and get stories like this direct to your inbox. Sign up here!
MAHA movement WINS: PepsiCo releases NEW Cheetos and Doritos alternatives amid RFK Jr.’s health crusade

Amid Robert F. Kennedy Jr.’s initiative to make our food healthy again, one major company has announced some new products stripped down to the “fundamentals” to please the MAHA crowd.
PepsiCo announced Thursday that it has developed a new line of Cheetos and Doritos without the artificial petroleum-based dyes — the ingredients that give them their intense color.
‘NKD is an additive option, not a replacement, introduced to meet consumer demand.’
While the old recipes are not going anywhere, PepsiCo announced the release of a new collection of snacks called Simply NKD, notably without the artificial dyes.
The press release boasts that the development process for these new snacks “came to life in just eight weeks.”
RELATED: MAHA agenda scores major win with announcement from food giant
 Photo Illustration by Justin Sullivan/Getty Images
Photo Illustration by Justin Sullivan/Getty Images
“Rest assured, our iconic Cheetos and Doritos remain unchanged. NKD is an additive option, not a replacement, introduced to meet consumer demand,” says Rachel Ferdinando, CEO of PepsiCo Foods U.S. “This move underscores our commitment to flavor leadership, demonstrating that our taste remains strong even without visual cues. As part of our broader transformation, we are expanding choices while still protecting our iconic brands. More choices, same flavor, same brand power.”
The new chips come in familiar flavors. Doritos will have Cool Ranch and Nacho Cheese, and Cheetos will have Puffs and Flamin’ Hot.
The new snacks appear to be a pale yellow, not the vibrant colors of the old recipe.
While the new Simply NKD varieties are supposed to taste like the originals, they will have shorter ingredient lists. As the AP reported, the new products do not have flavor-enhancing ingredients disodium inosinate and disodium guanylate, for example.
PepsiCo joins a growing list of companies that have removed or begun to phase out artificial ingredients from their foods lately. In-N-Out, Tyson Foods, Kraft Heinz, and McCormick have made similar commitments previously, to name a few.
These commitments come after the Food and Drug Administration banned Red No. 3 just before the beginning of Trump’s second term.
In response to a request for comment, HHS referred Blaze News to Robert F. Kennedy Jr.’s July 19, 2025, celebration of PepsiCo’s previous announcement that it planned to remove artificial colors and flavors from Lay’s and Tostitos.
At the time, RFK said, “PepsiCo just announced it will eliminate artificial colors and flavors from Lay’s and Tostitos by year’s end — and expand the use of avocado and olive oil in place of canola and soybean oil. I urge every other food company to follow their lead and join the movement to Make America Healthy Again.”
The new chips will be available in stores on December 1.
Editor’s note: The headline of this article was edited after publication for clarity.
Like Blaze News? Bypass the censors, sign up for our newsletters, and get stories like this direct to your inbox. Sign up here!
Can leucovorin cure autism? Meet the moms determined to find out

A humble, decades-old folate compound — used not to fight cancer but to ease the side effects of chemotherapy — has become the latest flashpoint in America’s health wars.
On September 10, the Trump administration announced that the FDA would move toward approving leucovorin for children with cerebral folate deficiency, a rare metabolic disorder linked to autism in some cases. Supporters hailed it as long-overdue recognition of promising small studies; critics called it another example of the MAHA agenda politicizing science.
While bureaucrats and scientists bicker, families with real skin in the game tirelessly run their own experiments and share their results, hoping the science will eventually catch up.
The debate since has been fierce, with professional groups such as the American Academy of Pediatrics advising against the off-label use of leucovorin for autism, warning that the evidence remains preliminary — while prominent physicians call for larger, biomarker-guided trials to confirm what early studies suggest.
A parent’s love
All parties insist their motives are pure, but this latest skirmish is a reminder of how tangled those motives can be. What drives the people and institutions pushing medical science forward is often a sincere desire to help people, yes — mixed in with ambition, rivalry, financial interest, and the unspoken urge to be the one who’s right.
But there’s another force at work here, deeper and simpler, and it tends to override all the rest: a parent’s love for a child.
This is the same love that kept the parents of children with cystic fibrosis pushing to understand a condition doctors considered hopeless, or that led a Hollywood father to resurrect a forgotten epilepsy therapy to help his son. And now it’s the force animating hundreds of parents who believe a decades-old folate compound has literally given their autistic children a voice.
While bureaucrats and scientists bicker, families with real skin in the game tirelessly run their own experiments and share their results, hoping the science will eventually catch up.
Even before the FDA signaled approval of leucovorin for cerebral folate deficiency — a rare metabolic disorder with links to autism — parents have been sharing reports of progress with the drug on Reddit forums and in Facebook groups to share anecdotal reports of progress. A few families have also told their stories in clinic-produced or news-segment videos.
A treatment’s hope
Leucovorin, also called folinic acid, is a bioactive form of folate. It’s been used for decades to “rescue” patients from high-dose chemotherapy. In autism, it’s being repurposed to bypass what some researchers call a “folate transport blockade.”
Up to 70% of autistic children in certain studies test positive for folate receptor alpha autoantibodies — immune proteins that prevent folate from reaching the brain. The result: cerebral folate deficiency. High-dose folinic acid appears to restore that supply, sometimes with striking behavioral effects.
Dr. Richard Frye, a pediatric neurologist at Phoenix Children’s Hospital, led one of the first controlled trials in 2016. His team found improved verbal communication in FRAA-positive children treated with leucovorin. Later case studies described language bursts, better eye contact, and calmer affect.
RELATED: Tylenol fights autism claims, slams proposed FDA warning label as ‘unsupported’ by science
 Photo by ISSAM AHMED/AFP via Getty Images
Photo by ISSAM AHMED/AFP via Getty Images
From ‘no words’ to the Pledge of Allegiance
The parents themselves provide more affecting testimony. Carolyn Connor’s son Mason was 1 when she realized something was amiss: “He wasn’t talking. No language. No words.”
When their pediatrician downplayed this lag in development as typical in boys, she and her husband began doing their own research, which led them to Frye. Three days after starting leucovorin, Mason spoke his first words.
Now 6, he continues to take the medication, and continues to thrive.
Beth Ann Kersse’s daughter was diagnosed with autism at age 3. “In her vocabulary she had about three or four words,” Kersse said in a video uploaded by Washington, D.C.-based Potomac Psychiatry.
“But she didn’t call me ‘Mom.’ She kind of would point at me,” she added.
That’s when Kersse and her husband began exploring leucovorin. Two years later, Kersse describes her almost 5-year-old daughter’s transformation as “incredible.”
“The other day she stood up and put her hand over her heart, and she recited the Pledge of Allegiance, and we were just like, OK … I didn’t know we knew that. … She’s able to have a full conversation; she can tell us how she’s feeling.”
Late last month, Nebraska pediatrician Dr. Phil Boucher posted a case study detailing how a 3.5-year-old autistic girl responded to leucovin treatment, citing texts from her mother reporting that she was “blown away” by the changes she observed:
She is starting to consistently look at people when they call her name. … She’s becoming more interested in her little sister. … She also has started taking some of the baby dolls that we have and has been covering them up with a blanket, giving them a kiss, and saying, “Night night.”
As Boucher is careful to point out, anecdotal success stories like these don’t prove the drug works. But to those experiencing the improvement firsthand, they’re a promising sign that a simple, inexpensive vitamin derivative can do what years of therapy can’t.
And if this promise does indeed bear fruit, leucovorin treatment will be the latest of many homegrown revolutions in medical care spearheaded by determined mothers and fathers unwilling to wait for consensus.
search
categories
Archives
navigation
Recent posts
- Maine Democrat Graham Platner Vows To Work With ‘Ron Paul,’ Thomas Massie if Elected to Senate: ‘Very Much Aligned’ April 13, 2026
- Dobol B TV Livestream: April 13, 2026 April 13, 2026
- LIVE UPDATES: Conflict in the Middle East (April 13, 2026) April 13, 2026
- Lebanon PM says he is working to get Israeli troop withdrawal April 13, 2026
- US to begin blockade of Iranian ports Monday –military April 13, 2026
- Hungary”s Orban concedes landmark defeat to centre-right opposition April 13, 2026
- Iran in crisis as US talks collapse, Mojtaba’s ‘mafia’ regime blocks Khamenei burial: analyst April 13, 2026







